Sirolimus vs Paclitaxel balloons for femoropopliteal disease: results from the SIRONA Trial
Selected in JACC by Cédric Lacoste
Sirolimus and paclitaxel drug-coated balloons (DCBs) are transforming the treatment of femoropopliteal artery disease. But which one delivers better vessel patency and clinical outcomes?
The SIRONA randomised non-inferiority trial directly compared sirolimus-coated balloons (SCBs) with paclitaxel-coated balloons (PCBs) in patients with symptomatic femoropopliteal lesions.
With 12-month follow-up, the study evaluated primary vessel patency and a composite clinical endpoint including revascularisation, major limb amputation, and device-related mortality, offering new insights for clinicians seeking effective, safe, and alternative endovascular options.
References:
Authors: Ulf Teichgräber, Maja Ingwersen, Thomas Lehmann, Stephanie Platzer, Ina Kunstmann, Sabine Steiner, Andrej Schmidt, Tim Wittig, Thomas Zeller, Marcus Thieme, Hans Krankenberg, Martin Werner, Marianne Brodmann, Nasser Malyar, Stefan Müller-Hülsbeck, Luca de Paoli, Oliver Schlager, Martin Austermann, Gunnar Tepe, Christian Erbel, Wulf Ito, Rainer Waßmer, Lars Maiwald, René Aschenbach, Christian Wissgott, Martin Andrassy, Florian Wolf, Ulrich Beschorner, Maciej Pech, Konstantinos Katsanos, Dierk Scheinert, and the SIRONA Study Group
Reference: J Am Coll Cardiol . 2026 Feb 18:S0735-1097(25)10433-6
DOI: 10.1016/j.jacc.2025.12.017
Read the abstractObjective:
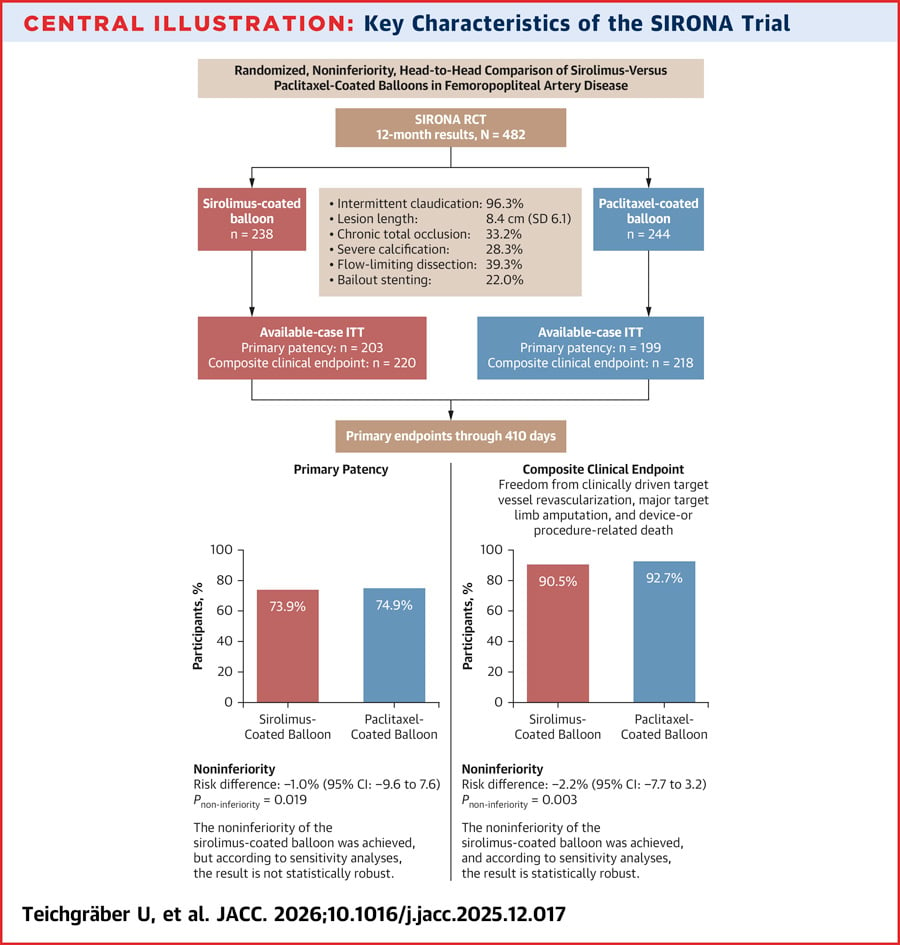
The aim is to investigate whether SIROLIMUS–coated balloon angioplasty achieves non-inferior primary vessel patency and clinical efficacy and safety compared with PACLITAXEL–coated balloon angioplasty.
Study and population:
Randomised, controlled, non-inferiority trial in patient with symptomatic femoropopliteal artery.
Endpoints:
- Primary efficacy endpoint: primary vessel patency at 12 months.
- Primary clinical endpoint: composite of freedom from clinically driven target vessel revascularisation, major target limb amputation, and device- or procedure-related death at 12 months.
Key Findings:
- Primary patency: 150/203 (73.9%) with SIROLIMUS vs 149/199 (74.9%) with PACLITAXEL (risk difference: -1.0%; 95% CI: -9.6% to 7.6%; P_non-inferiority = 0.019). Statistically non-robust.
- Primary clinical endpoint: 199/220 (90.5%) with SCBs vs 202/218 (92.7%) with PCBs (risk difference: -2.2%; 95% CI: -7.7% to 3.2%; P_non-inferiority = 0.003). Confirmed by sensitivity analyses.
Get the latest clinical cases and breaking news delivered straight to your inbox!


Conclusion:
Among patients with femoropopliteal artery disease, SIROLIMUS demonstrated non-inferior clinically important outcomes.
While non-inferiority for primary patency was not statistically robust, SCBs may still be a valuable alternative to PACLITAXEL-coated balloons.
Discussion :
No cost comparison or detailed safety evaluation; long-term follow-up in a larger population is needed.